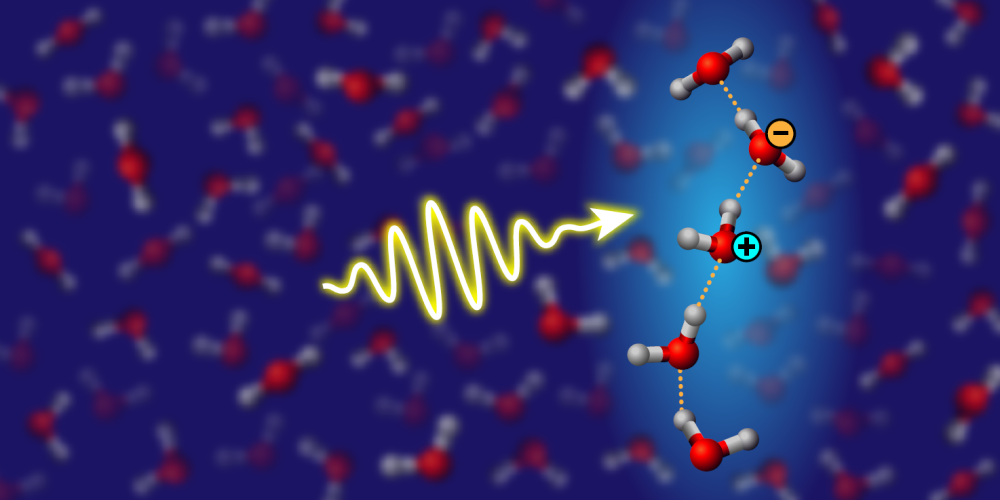
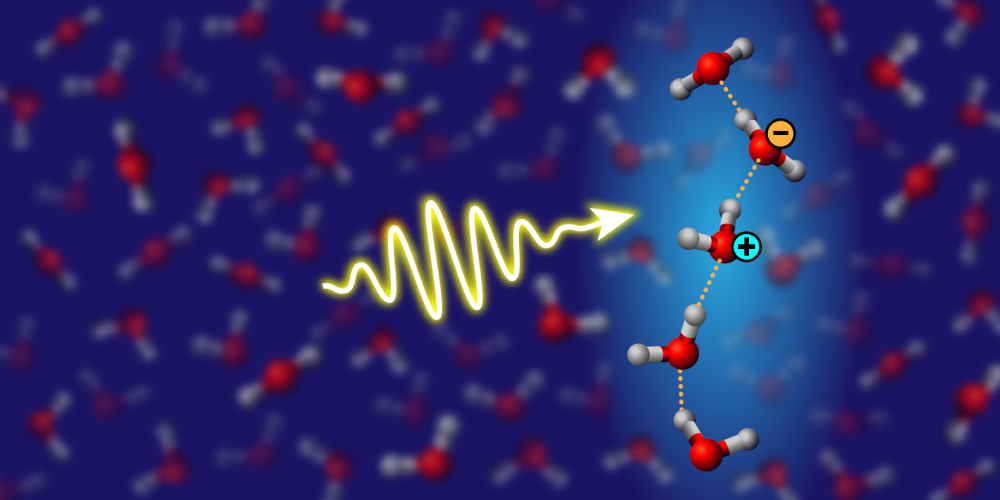
Shedding Light on Water Wires
A hydrogen bond is an electrostatic attraction between molecules, mediated by a positively charged hydrogen atom. This relatively weak bond—first theoretically described in 1920 [1]—is the fundamental binding force in water, explaining many of this liquid’s unique properties, such as its high surface tension and unique attributes as a solvent. Within the hydrogen-bonded network connecting H2O molecules in water and ice, stronger hydrogen bonds can form short-lived chains, called water wires, but the effect of these linear structures is still debated. Molecular simulations suggest that water wires provide important pathways for proton transfer, but direct observational evidence has been lacking. Fujie Tang from Temple University in Pennsylvania and colleagues propose a way to measure the presence of water wires [2] (Fig. 1). Through rigorous electronic-structure calculations and molecular-dynamics simulations, the researchers show that elusive water wires can be detected in bulk water and ice by measuring light absorption in the UV-to-visible range. The technique could be used to study how water-wire behavior changes under different spatial and thermal conditions.
The transport of protons (H+ ions) is an important mechanism behind water’s properties. Proton transfer along hydrogen-bond pathways enables electrical conduction in ice and acid–base chemistry in water. In biology, water-based proton transfer is a fundamental molecular mechanism governing the functioning of several processes, such as chemical transport through membrane channels. One way that such proton transfer might be enhanced is through water wires. Previous work with first-principles molecular-dynamics simulations showed that protons should preferentially hop along wires composed of a few suitably aligned and tightly bonded water molecules [3]. Confirming this picture, however, has been difficult. While vibrational spectroscopy using infrared light is one of the most common tools for the experimental characterization of the structure and dynamics of hydrogen-bonded systems, the presence of molecular wires in bulk water is hard to detect because of their fleeting nature and the lack of specific vibrational fingerprints.
To obtain a potential water-wire signature, Tang and colleagues turned to optical spectroscopy. By combining molecular-dynamics simulations and calculations of optical spectra based on many-body perturbation theory, they identified a clear spectroscopic fingerprint of molecular wires. Specifically, they showed that the “charge-transfer exciton” peak at 8 eV in the UV absorption spectrum of water stems from collective excitations in hydrogen-bonded water wires. A charge-transfer exciton is a special type of electronic excitation, where an electron on one water molecule interacts with a positively charged “hole” localized on another molecule. The strength of the hydrogen bond directly influences the degree of charge transfer between molecules, with stronger hydrogen bonds facilitating greater charge separation and providing stronger optical absorption. Interestingly, Tang and colleagues found that the intensity of the peak can be linked to the length and stability of the water wires.
The calculations by Tang and colleagues combined several state-of-the-art techniques, including the quantum description of the nuclear motion using path-integral molecular dynamics. These techniques exploit artificial intelligence to overcome the computational cost of quantum simulations and extend their reach to unprecedented timescales and size scales [4–6]. The researchers obtained good agreement between computed and measured absorption spectra for water and ice, thus validating their approach and showing the importance of using accurate many-body techniques to describe light–matter interaction in hydrogen-bonded systems.
To identify which features in their computed spectrum are due to the presence of water wires, the researchers performed simulations for three different phases: water, proton-disordered ice, and proton-ordered ice. Proton-disordered ice is the familiar solid in our cold drinks; it has a crystalline structure but with randomly oriented hydrogen atoms. Proton-ordered ice has its hydrogens lined up, but it occurs only at cryogenic temperatures. Effects from water wires should be more prominent in proton-ordered ice and weaker in liquid water. Indeed, the researchers showed that the intensity of the charge-transfer-exciton peak is highest for the proton-ordered ice model, where the water wires extend the length of the material. The peak intensity is lower for the proton-disordered ice model, where water wires are composed of just a few water molecules. And the signal is weaker in liquid water, where the wires dynamically form and break over time.
These results suggest a number of possible experiments. For example, researchers could estimate the length and stability of water wires by monitoring the spectral features of a water sample while varying one of the system parameters, such as temperature, pressure, confinement volume, or solution composition. Researchers could also use UV-visible spectroscopy to gain a better understanding of proton transfer in chemical reactions (for example, acid–base regulation and enzyme catalysis) and in biological processes (for example, photosynthesis and mitochondrial respiration) [7]. These investigations might also uncover new insights into some of water’s puzzling anomalies, such as the density maximum at 4 °C and the compressibility minimum at 46 °C. One aspect of water that is currently under debate is whether there is a liquid–liquid phase transition in the deep supercooled region [8]. Researchers might be able to resolve this issue by using the newly identified spectral markers to search for the coexistence of two liquid states with different densities.
The promises of optical spectroscopy to unravel hydrogen-bonding configurations are clearly not limited to water and biological systems. The UV-visible detection of excitonic peaks may be used for a myriad of other systems, including aqueous solutions and hydrogen-bonded liquids in contact with surfaces. It can also be used for the characterization and optimization of energy-conversion systems, such as organic light-emitting devices (OLEDs) [9] and fuel cells [10], where hydrogen bonding, excitonic transitions, and proton transfer play a key role.
References
- W. M. Latimer and W. H. Rodebush, “Polarity and ionization from the standpoint of the Lewis theory of valence,” J. Am. Chem. Soc. 42, 1419 (1920).
- F. Tang et al., “Optical absorption spectroscopy probes water wire and its ordering in a hydrogen-bond network,” Phys. Rev. X 15, 011048 (2025).
- A. Hassanali et al., “On the recombination of hydronium and hydroxide ions in water,” Proc. Natl. Acad. Sci. U.S.A. 108, 20410 (2011).
- J. Behler and M. Parrinello, “Generalized neural-network representation of high-dimensional potential-energy surfaces,” Phys. Rev. Lett. 98, 146401 (2007).
- L. Zhang et al., “Active learning of uniformly accurate interatomic potentials for materials simulation,” Phys. Rev. Mater. 3, 023804 (2019).
- M. L. Berrens et al., “Nuclear quantum effects on the electronic structure of water and ice,” J. Phys. Chem. Lett. 15, 6818 (2024).
- M. Wikström et al., “New perspectives on proton pumping in cellular respiration,” Chem. Rev. 115, 2196 (2015).
- P. Gallo et al., “Water: A tale of two liquids,” Chem. Rev. 116, 7463 (2016).
- J. Sun et al., “Charge-transfer exciton manipulation based on hydrogen bond for efficient white thermally activated delayed fluorescence,” Adv. Funct. Mater. 30 (2019).
- K.-D. Kreuer et al., “Transport in proton conductors for fuel-cell applications: simulations, elementary reactions, and phenomenology,” Chem. Rev. 104, 4637 (2004).